N2 + O2 = N2O - Balanced Chemical Equation

Reactions Balance the equation Mg + HNO3 -> Mg(NO3)2 + N2O + H20 0 +5 +2 +5 +1

Solved The decomposition of N2O has the following overall

How to Balance N2 + O2 = N2O3 (Nitrogen gas + Oxygen gas)

Consider this reaction occurring at 298 K: N2O( g) + NO2( g) ∆ 3

SOLVED: Balancing Chemical Equations LEVEL 1 - NOVICE H2 + O2 -> H2O C + S8 -> CS2 NaO2 -> Na2O2 N2 + O2 -> N2O H2 + O2 -> H2O2 Na +
Reaction between N2 and O2 takes place as follows: 4N2 (g) + O2 (g) 2N2O (g) If a mixture of 0.482 mol N2 and 0.933 mol O2 is placed in a 10L
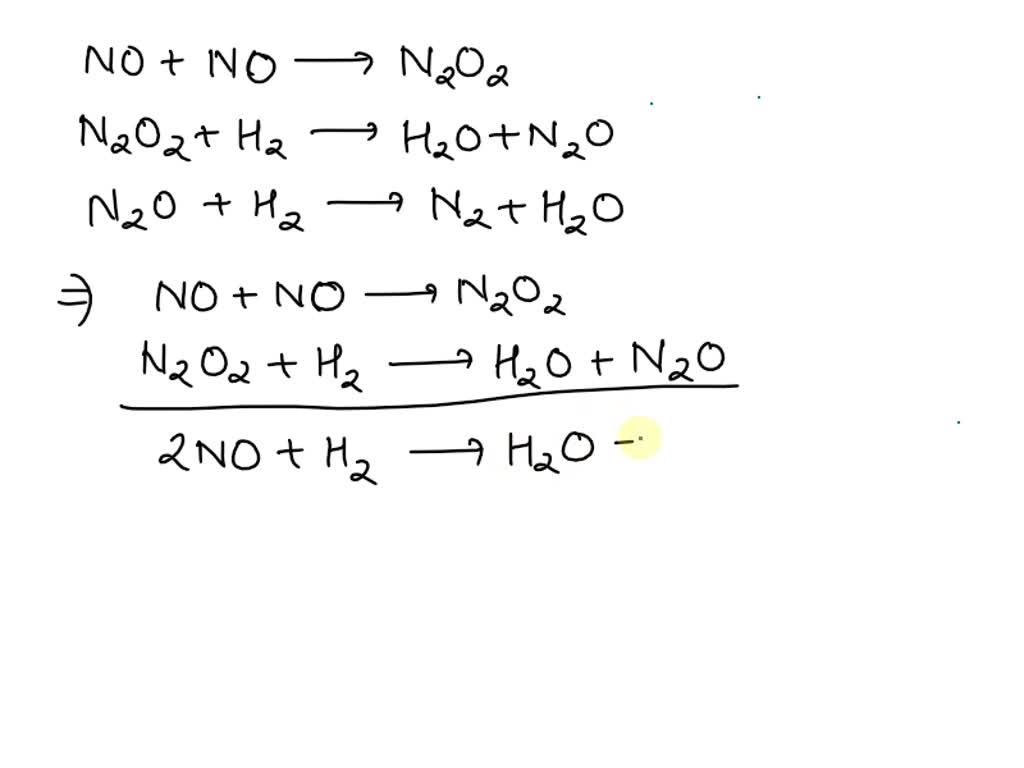
SOLVED: The following sequence of elementary steps is a proposed mechanism for a reaction: NO + NO -> N2O2 N2O2 + H2 -> H2O + N2O N2O + H2 -> N2 +

Solved Q2- Nitrous oxide (N2O) decomposes at 600∘C according

N2+O2+H2O=HNO3 balance the chemical equation @mydocumentary838. n2+o2+h2o=hno3 balance the equation

N2+O2 =NO2 Balanced Equation